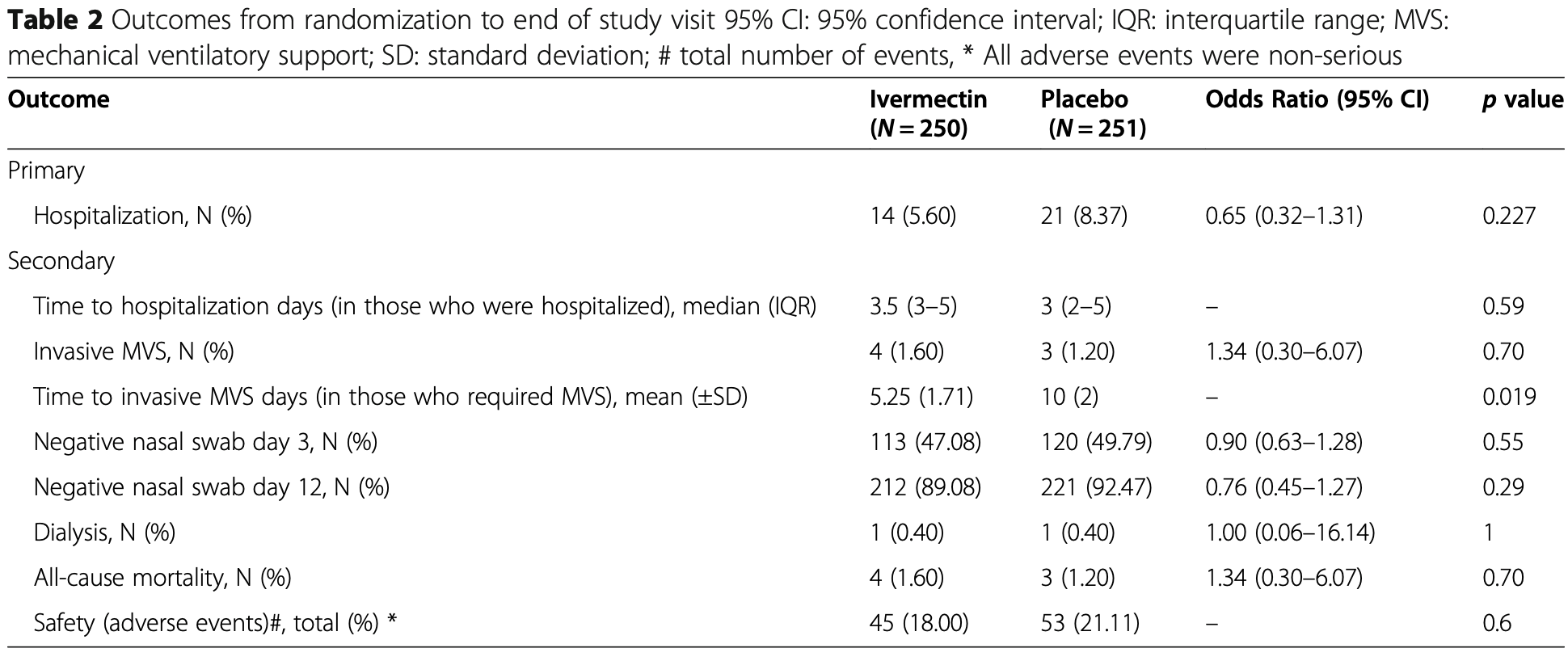
RCT with 501 relatively low-risk outpatients in Argentina showing
hospitalization OR 0.65 [0.32-1.31].
With only 7% hospitalization, this trial is underpowered. The trial primarily
includes low-risk patients that recover quickly without treatment, leaving
minimal room for improvement with treatment. 74 patients had symptoms for >= 7
days. Among the 7 patients requiring ventilation, authors note that the
earlier requirement in the ivermectin group may be due to those patients
having higher severity at baseline. However, authors know the answer to this -
it is unclear why it is not reported. There were more adverse events in the
placebo group than the ivermectin group, suggesting a possible issue with
dispensing or non-trial medication usage. 25+% of patients were hospitalized
within 2/3 days for the placebo/treatment groups (Figure S2).
The companion prophylaxis study
IVERCOR PREP has
reported results in the press and an online presentation
lanacion.com.ar, ,
however these results have not yet been formally published. The prophylaxis
study results are very positive and statistically significant, and would be
expected to receive priority publication due to the predicted impact on the
pandemic and confirmation of previous prophylaxis studies. The lack of formal
publication suggests a negative publication bias that may be due to
politicization in the authors' location.
Authors pre-specify multivariate analysis but do not present
it, however multivariate analysis could significantly change the results.
Consider for example if just a few extra patients in the ivermectin group
were in severe condition based on baseline SpO2. The lower mean SpO2 in the
ivermectin group, and the shorter time to ventilation, are consistent with
this being the case. Additionally, there are 14% more male patients in the
ivermectin group.
An extremely large percentage of patients (55%) were excluded
based on ivermectin use in the last 7 days. However, ivermectin may retain
efficacy much longer (for example antiparasitic activity may persist for
months
Canga). A significant number of patients may also
misrepresent their prior and future usage — the population is clearly
aware of ivermectin, and patients with progressing disease may be motivated
to take it, knowing that they may be in the control group. Another report
states that 12,000 patients were excluded for recent use of ivermectin
scidev.net).
RCTs have a fundamental bias against finding an effect for
interventions that are widely available — patients that believe they
need treatment are more likely to decline participation and take the
intervention
Yeh, i.e., RCTs are more likely to enroll low-risk
participants that do not need treatment to recover (this does not apply to
the typical pharmaceutical trial of a new drug that is otherwise
unavailable). This trial was run in a community where ivermectin was very
widely known and used.
|
risk of death, 33.5% higher, RR 1.33, p = 0.70, treatment 4 of 250 (1.6%), control 3 of 251 (1.2%), odds ratio converted to relative risk.
|
|
risk of mechanical ventilation, 33.5% higher, RR 1.33, p = 0.70, treatment 4 of 250 (1.6%), control 3 of 251 (1.2%), odds ratio converted to relative risk.
|
|
risk of hospitalization, 33.0% lower, RR 0.67, p = 0.23, treatment 14 of 250 (5.6%), control 21 of 251 (8.4%), NNT 36, odds ratio converted to relative risk, primary outcome.
|
|
risk of no viral clearance, 5.0% higher, RR 1.05, p = 0.55, treatment 137 of 250 (54.8%), control 131 of 251 (52.2%), inverted to make RR<1 favor treatment, odds ratio converted to relative risk, day 3.
|
|
risk of no viral clearance, 26.8% higher, RR 1.27, p = 0.29, treatment 38 of 250 (15.2%), control 30 of 251 (12.0%), inverted to make RR<1 favor treatment, odds ratio converted to relative risk, day 12.
|
|
Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates
|
Vallejos et al., 2 Jul 2021, Double Blind Randomized Controlled Trial, Argentina, peer-reviewed, 29 authors, study period 19 August, 2020 - 22 February, 2021, average treatment delay 4.0 days, dosage 12mg days 1-2, <80kg 12mg, 80-110kg 18mg, >110kg 24mg.
Ivermectin to prevent hospitalizations in patients with COVID-19 (IVERCOR-COVID19) a randomized, double-blind, placebo-controlled trial
Julio Vallejos, Rodrigo Zoni, María Bangher, Silvina Villamandos, Angelina Bobadilla, Fabian Plano, Claudia Campias, Evangelina Chaparro Campias, Maria Fernanda Medina, Fernando Achinelli, Hector Andres Guglielmone, Jorge Ojeda, Diego Farizano Salazar, Gerardo Andino, Pablo Kawerin, Silvana Dellamea, Antonia Cristina Aquino, Victor Flores, Carolina N Martemucci, Silvina Maria Martinez, Juan Emanuel Segovia, Paola Itati Reynoso, Noelia Carolina Sosa, Mariana Elizabeth Robledo, Joaquina Maria Guarrochena, Maria Mercedes Vernengo, Natalia Ruiz Diaz, Elba Meza, María Gabriela Aguirre
BMC Infectious Diseases, doi:10.1186/s12879-021-06348-5
Background: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) has changed our lives. The scientific community has been investigating re-purposed treatments to prevent disease progression in coronavirus disease patients. Objective: To determine whether ivermectin treatment can prevent hospitalization in individuals with early COVID-19. Design, setting and participants: A randomized, double-blind, placebo-controlled study was conducted in nonhospitalized individuals with COVID-19 in Corrientes, Argentina. Patients with SARS-CoV-2 positive nasal swabs were contacted within 48 h by telephone to invite them to participate. The trial randomized 501 patients between August 19th 2020 and February 22nd 2021. Intervention: Patients were randomized to ivermectin (N = 250) or placebo (N = 251) arms in a staggered dose, according to the patient's weight, for 2 days. Main outcomes and measures: The efficacy of ivermectin to prevent hospitalizations was evaluated as primary outcome. We evaluated secondary outcomes in relationship to safety and other efficacy end points.
Supplementary Information The online version contains supplementary material available at https://doi. org/10.1186/s12879-021-06348-5.
Additional file 1.
Authors' contributions
Declarations Ethics approval and consent to participate study authorized by Health Sciences Research Bioethics Committee (HSRBC) of the National University of the Northeast (UNNE) Faculty of Medicine, Argentina (Resolution 21/20 on August 17th, 2020). The consent to participate in the trial was approved by HSRBC of the UNNE. The study was performance in accordance with the Declaration of Helsinki and all methods were performed in accordance with the relevant guidelines and regulations. Informed consent was obtained from all participants prior to the study. The study has been supervised by a Steering Committee and Safety Committee.
Consent for publication Not Applicable.
Competing interests The authors declare that they have no competing interests.
Publisher's Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Ahmed, Hany, Youssef, Hany, Hafez et al., Efficacy and safety of ivermectin for treatment and prophylaxis of COVID-19 pandemic,
doi:10.21203/rs.3.rs-100956/v1Al-Abdouh, Bizanti, Barbarawi, Remdesivir for the treatment of COVID-19: a systematic review and meta-analysis of randomized controlled trials, Contemp Clin Trials,
doi:10.1016/j.cct.2021.106272Babalola, Bode, Ajayi, Ivermectin shows clinical benefits in mild to moderate COVID19: a randomised controlled double blind dose response study in Lagos. medRxiv,
doi:10.1101/2021.01.05.21249131Bartoli, Gabrielli, Alicandro, Nascimbeni, Andreoni, COVID-19 treatment options: a difficult journey between failed attempts and experimental drugs, Intern Emerg Med,
doi:10.1007/s11739-020-02569-9Beigel, Tomashek, Dodd, Mehta, Zingman et al., Remdesivir for the treatment of Covid-19 -final report, N Engl J Med,
doi:10.1056/NEJMoa2007764Camprubí, Almuedo-Riera, Martí-Soler, Lack of efficacy of standard doses of ivermectin in severe COVID-19 patients, PLoS One
Chaccour, Casellas, Blanco-Di Matteo, The effect of early treatment with ivermectin on viral load, symptoms and humoral response in patients with non-severe COVID-19: a pilot, double-blind, placebocontrolled, randomized clinical trial, EClinicalMedicine,
doi:10.1016/j.eclinm.2020.100720Gotz, Magar, Dornfeld, Influenza a viruses escape from MxA restriction at the expense of efficient nuclear vRNP import, Sci Rep,
doi:10.1038/srep23138Hashim, Maulood, Rasheed, Dhurgham, Fatak et al., Controlled randomized clinical trial on using ivermectin with doxycycline for treating COVID-19 patients in Baghdad, Iraq medRxiv Preprint,
doi:10.1101/2020.10.26.20219345Khan, Khan, Debnath, Nath, Mahtab et al., Ivermectin treatment may improve the prognosis of patients with COVID
Lai, Liu, Wang, Wang, Hsueh et al., Asymptomatic carrier state, acute respiratory disease, and pneumonia due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): facts and myths, J Microbiol Immunol Infect,
doi:10.1016/j.jmii.2020.02.012Libster, Marc, Wappner, Coviello, Bianchi et al., Early high-titer plasma therapy to prevent severe Covid-19 in older adults, N Engl J Med,
doi:10.1056/NEJMoa2033700López-Medina, López, Hurtado, Dávalos, Ramirez et al., Effect of ivermectin on time to resolution of symptoms among adults with mild COVID-19: a randomized clinical trial, JAMA,
doi:10.1001/jama.2021.3071Mahmud, Rahman, Alam, Ahmed, Kabir et al., Ivermectin in combination with doxycycline for treating COVID-19 symptoms: a randomized trial, J Int Med Res,
doi:10.1177/03000605211013550Mega, Latin America's embrace of an unproven COVID treatment is hindering drug trials, Nature
Navarro, Camprubí, Requena-Méndez, Buonfrate, Giorli et al., Safety of high-dose ivermectin: a systematic review and metaanalysis, J Antimicrob Chemother,
doi:10.1093/jac/dkz524Padhy, Mohanty, Das, Meher, Therapeutic potential of ivermectin as add on treatment in COVID 19: a systematic review and meta-analysis, J Pharm Pharm Sci,
doi:10.18433/jpps31457Ross, Clemens, Sumiya, A five-day course of ivermectin for the treatment of COVID-19 may reduce the duration of illness, Int J Infect Dis,
doi:10.1016/j.ijid.2020.11.191Salama, Han, Yau, Reiss, Kramer et al., Tocilizumab in patients hospitalized with Covid-19 pneumonia, N Engl J Med,
doi:10.1056/NEJMoa2030340Simonovich, Pratx, Scibona, Beruto, Vallone et al., A randomized trial of convalescent plasma in Covid-19 severe pneumonia, N Engl J Med,
doi:10.1056/NEJMoa2031304Tay, Fraser, Chan, Nuclear localization of dengue virus (DENV) 1-non-structural protein 5; protection against all 4 DENV serotypes by the inhibitor Ivermectin, Antivir Res,
doi:10.1016/j.antiviral.2013.06.002Vallejos, Zoni, Bangher, Villamandos, Bobadilla et al., Ivermectin to prevent hospitalizations in patients with COVID-19 (IVERCOR-COVID19): a structured summary of a study protocol for a randomized controlled trial, Trials,
doi:10.1186/s13063-020-04813-1Wagstaff, Sivakumaran, Heaton, Harrich, Jans, Ivermectin is a specific inhibitor of importin alpha/betamediated nuclear import able to inhibit replication of HIV-1 and dengue virus, Biochem J,
doi:10.1042/BJ20120150Zhou, Yu, Du, Clinical course and risk factors for mortality of adult inpatients with COVID-19 inWuhan, China: a retrospective cohort study, Lancet,
doi:10.1016/S0140-6736(20)30566-3